NC Standards
Chm.1.2 Understand the bonding that occurs in simple compounds in terms of bond type, strength, and properties.
Chm.1.2.1 Compare (qualitatively) the relative strengths of ionic, covalent, and metallic bonds.
Describe metallic bonds: “metal ions plus ‘sea’ of mobile electrons”.
Describe how ions are formed and which arrangements are stable (filled d-level, or half-filled d-level).
Appropriately use the term cation as a positively charged ion and anion as negatively charged ion.
Predict ionic charges for representative elements based on valence electrons.
Apply the concept that sharing electrons form a covalent compound that is a stable (inert gas) arrangement.
Draw Lewis (dot diagram) structures for simple compounds and diatomic elements indicating single, double or triple bonds.
Chm.1.2.2 Infer the type of bond and chemical formula formed between atoms.
Determine that a bond is predominately ionic by the location of the atoms on the Periodic Table (metals combined with nonmetals) or when Delta EN > 1.7.
Determine that a bond is predominately covalent by the location of the atoms on the Periodic Table (nonmetals combined with nonmetals) or when Delta EN < 1.7.
Predict chemical formulas of compounds using Lewis structures.
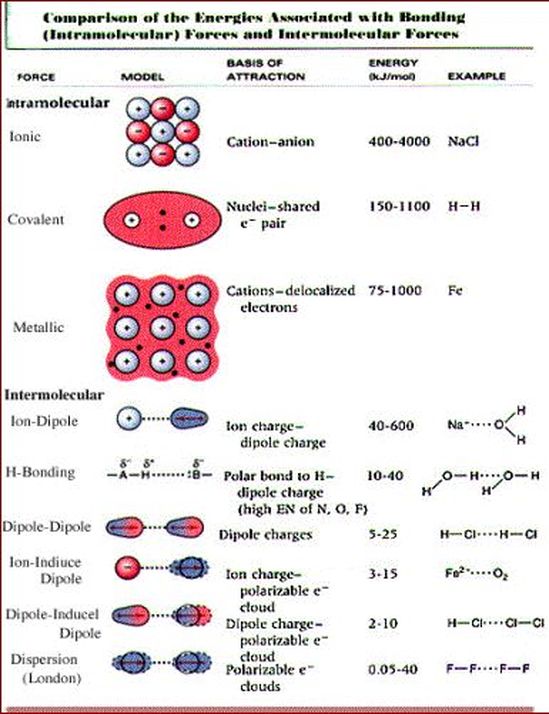
Chm.1.2.3 Compare inter- and intra- particle forces.
Explain why intermolecular forces are weaker than ionic, covalent or metallic bonds.
Explain why hydrogen bonds are stronger than dipole-dipole forces which are stronger than dispersion forces.
Apply the relationship between bond energy and length of single, double, and triple bonds (conceptual, no numbers).
Describe intermolecular forces for molecular compounds. o H-bond as attraction between molecules when H is bonded to O, N, or F. Dipole-dipole attractions between polar molecules. o London dispersion forces (electrons of one molecule attracted to nucleus of another molecule) – i.e. liquefied inert gases. o Relative strengths (H>dipole>London/van der Waals).
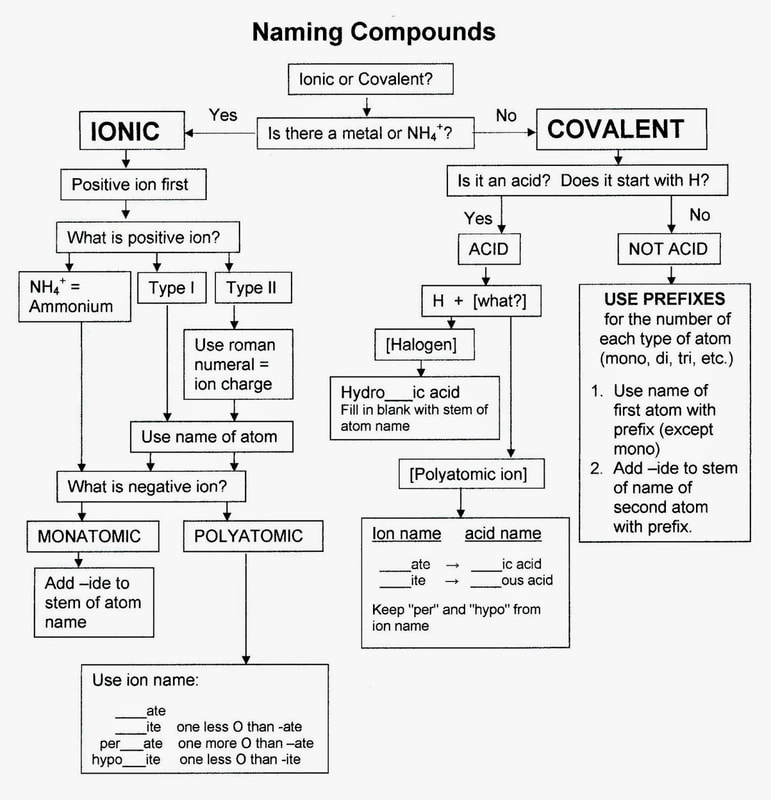
Chm.1.2.4 Interpret the name and formula of compounds using IUPAC convention.
Write binary compounds of two nonmetals: use Greek prefixes (di-, tri-, tetra-, …).
Write binary compounds of metal/nonmetal*.
Write ternary compounds (polyatomic ions)* using the polyatomic ions on the reference table. Write, with charges, these polyatomic ions: nitrate, sulfate, carbonate, acetate, and ammonium.
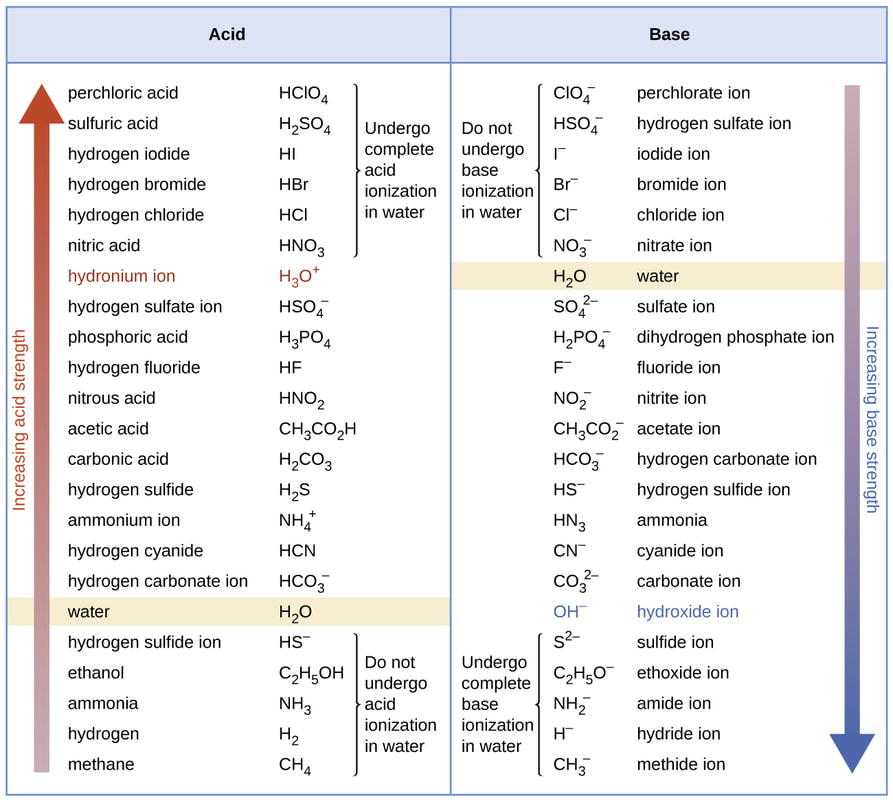
Know names and formulas for these common laboratory acids: HCl, HNO3, H2SO4, HC2H3O2, (CH3COOH) *The Stock system is the correct IUPAC convention for inorganic nomenclature.
Chm.1.2.5 Compare the properties of ionic, covalent, metallic, and network compounds.
Explain how ionic bonding in compounds determines their characteristics: high MP, high BP, brittle, and high electrical conductivity either in molten state or in aqueous solution.
Explain how covalent bonding in compounds determines their characteristics: low MP, low BP, poor electrical conductivity, polar nature, etc.
Explain how metallic bonding determines the characteristics of metals: high MP, high BP, high conductivity, malleability, ductility, and luster.
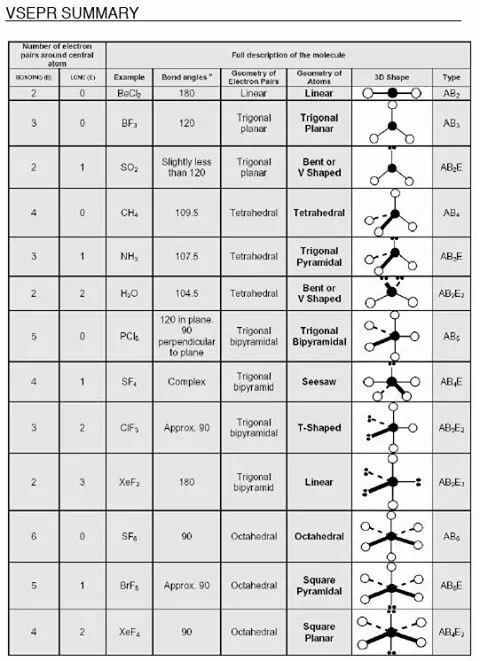
Apply Valence Shell Electron Pair Repulsion Theory (VSEPR) for these electron pair geometries and molecular geometries, and bond angles - Electron pair - Molecular (bond angle); Linear framework – linear; Trigonal planar framework– trigonal planar, bent; Tetrahedral framework– tetrahedral, trigonal pyramidal, bent; Bond angles (include distorting effect of lone pair electrons – no specific angles, conceptually only).
Describe bond polarity. Polar/nonpolar molecules (relate to symmetry) ; relate polarity to solubility—“like dissolves like”.
Describe macromolecules and network solids: water (ice), graphite/diamond, polymers (PVC, nylon), proteins (hair, DNA) intermolecular structure as a class of molecules with unique properties.
Chm.1.2.1 Compare (qualitatively) the relative strengths of ionic, covalent, and metallic bonds.
Describe metallic bonds: “metal ions plus ‘sea’ of mobile electrons”.
Describe how ions are formed and which arrangements are stable (filled d-level, or half-filled d-level).
Appropriately use the term cation as a positively charged ion and anion as negatively charged ion.
Predict ionic charges for representative elements based on valence electrons.
Apply the concept that sharing electrons form a covalent compound that is a stable (inert gas) arrangement.
Draw Lewis (dot diagram) structures for simple compounds and diatomic elements indicating single, double or triple bonds.
Chm.1.2.2 Infer the type of bond and chemical formula formed between atoms.
Determine that a bond is predominately ionic by the location of the atoms on the Periodic Table (metals combined with nonmetals) or when Delta EN > 1.7.
Determine that a bond is predominately covalent by the location of the atoms on the Periodic Table (nonmetals combined with nonmetals) or when Delta EN < 1.7.
Predict chemical formulas of compounds using Lewis structures.
Chm.1.2.3 Compare inter- and intra- particle forces.
Explain why intermolecular forces are weaker than ionic, covalent or metallic bonds.
Explain why hydrogen bonds are stronger than dipole-dipole forces which are stronger than dispersion forces.
Apply the relationship between bond energy and length of single, double, and triple bonds (conceptual, no numbers).
Describe intermolecular forces for molecular compounds. o H-bond as attraction between molecules when H is bonded to O, N, or F. Dipole-dipole attractions between polar molecules. o London dispersion forces (electrons of one molecule attracted to nucleus of another molecule) – i.e. liquefied inert gases. o Relative strengths (H>dipole>London/van der Waals).
Chm.1.2.4 Interpret the name and formula of compounds using IUPAC convention.
Write binary compounds of two nonmetals: use Greek prefixes (di-, tri-, tetra-, …).
Write binary compounds of metal/nonmetal*.
Write ternary compounds (polyatomic ions)* using the polyatomic ions on the reference table. Write, with charges, these polyatomic ions: nitrate, sulfate, carbonate, acetate, and ammonium.
Know names and formulas for these common laboratory acids: HCl, HNO3, H2SO4, HC2H3O2, (CH3COOH) *The Stock system is the correct IUPAC convention for inorganic nomenclature.
Chm.1.2.5 Compare the properties of ionic, covalent, metallic, and network compounds.
Explain how ionic bonding in compounds determines their characteristics: high MP, high BP, brittle, and high electrical conductivity either in molten state or in aqueous solution.
Explain how covalent bonding in compounds determines their characteristics: low MP, low BP, poor electrical conductivity, polar nature, etc.
Explain how metallic bonding determines the characteristics of metals: high MP, high BP, high conductivity, malleability, ductility, and luster.
Apply Valence Shell Electron Pair Repulsion Theory (VSEPR) for these electron pair geometries and molecular geometries, and bond angles - Electron pair - Molecular (bond angle); Linear framework – linear; Trigonal planar framework– trigonal planar, bent; Tetrahedral framework– tetrahedral, trigonal pyramidal, bent; Bond angles (include distorting effect of lone pair electrons – no specific angles, conceptually only).
Describe bond polarity. Polar/nonpolar molecules (relate to symmetry) ; relate polarity to solubility—“like dissolves like”.
Describe macromolecules and network solids: water (ice), graphite/diamond, polymers (PVC, nylon), proteins (hair, DNA) intermolecular structure as a class of molecules with unique properties.
Vocabulary
ion
covalent bond
ionic bond
metallic bond
metallic (sea of mobile electrons)
valence electrons,
stable arrangement (inert gas)
octet rule
full and half-full sublevels
cation
anion
Lewis structure (dot diagram)
diatomic elements
single, double, and triple bonds
melting point
boiling point
brittle
ductile
malleable
conductivity
luster
molten state
aqueous solution
VSEPR theory
electron-pair geometry
molecular geometry
bond angles
linear
trigonal planar
bent
tetrahedral
trigonal pyramidal
bond polarity
polar
non-polar
symmetry
solubility
macro-molecule
network solid
graphite/diamond
polymers (PVC, nylon)
proteins (hair, DNA)
inter-molecular structure
covalent bond
ionic bond
metallic bond
metallic (sea of mobile electrons)
valence electrons,
stable arrangement (inert gas)
octet rule
full and half-full sublevels
cation
anion
Lewis structure (dot diagram)
diatomic elements
single, double, and triple bonds
melting point
boiling point
brittle
ductile
malleable
conductivity
luster
molten state
aqueous solution
VSEPR theory
electron-pair geometry
molecular geometry
bond angles
linear
trigonal planar
bent
tetrahedral
trigonal pyramidal
bond polarity
polar
non-polar
symmetry
solubility
macro-molecule
network solid
graphite/diamond
polymers (PVC, nylon)
proteins (hair, DNA)
inter-molecular structure
Types of Bonds
Learning Targets
I can describe the three types of bonds and how they form.
I can describe the characteristics of ionic, covalent, and metallic bonds.
I can predict the formula of a compound using Lewis structures.
I can describe the characteristics of ionic, covalent, and metallic bonds.
I can predict the formula of a compound using Lewis structures.
| bondingandproperties.key | |
| File Size: | 1421 kb |
| File Type: | key |
| 46electronegativityandbondpolarity.ppt | |
| File Size: | 484 kb |
| File Type: | ppt |
| lewis_dot_structures_for_compounds.ppt | |
| File Size: | 308 kb |
| File Type: | ppt |
| bondtype.docx | |
| File Size: | 11 kb |
| File Type: | docx |
| bonding_packet__1_.doc | |
| File Size: | 1824 kb |
| File Type: | doc |
| lewis_structures.doc | |
| File Size: | 24 kb |
| File Type: | doc |
Nomenclature
Learning Targets
I can predict the formula of a compound using Lew structures.
I can write names and charges for monatomic ions and polyatomic ions.
I can write names and charges for monatomic ions and polyatomic ions.
| naming_ions__1_.pptx | |
| File Size: | 47 kb |
| File Type: | pptx |
| naming_practice__1_.docx | |
| File Size: | 17 kb |
| File Type: | docx |
http://city.espora.co/compound-names/
Acids and Bases
Learning Targets
I can identify and name common acids and bases.
Acids & Bases Naming Practice
Write the formula or name for the 1st 10 questions in each chart.
| ws-_acid_nomenclature.doc | |
| File Size: | 50 kb |
| File Type: | doc |
Intermolecular Forces
Learning Targets
I can describe and compare the types of intermolecular forces for molecular compounds.
I can compare inter- and intra-molecular forces.
I can compare inter- and intra-molecular forces.
Intermolecular Forces Jigsaw
| intermolecular_forces.ppt | |
| File Size: | 318 kb |
| File Type: | ppt |
| compound_forces_word_map_2.docx | |
| File Size: | 69 kb |
| File Type: | docx |
Intermolecular vs Intramolecular Forces Webquest
| imf_webquest___graph_reading_activity_2017.docx | |
| File Size: | 44 kb |
| File Type: | docx |
https://www.pinterest.com/pin/805651820812756662/
VSEPR Theory and Polarity
Learning Targets
I can apply VSEPR theory to covalent compounds and predict molecular geometries.
https://www.pinterest.com/pin/434175220306021444/
VSEPR Theory Powerpoint
| polarity__1_.ppt | |
| File Size: | 319 kb |
| File Type: | ppt |
| vsepr___polarity_practice.docx | |
| File Size: | 13 kb |
| File Type: | docx |
| doc100112-10012012150156.pdf | |
| File Size: | 53 kb |
| File Type: | |