NC Standards
Chm.3.2 Understand solutions and the solution process.
Chm.3.2.1 Classify substances using the hydronium and hydroxide concentrations.
• Distinguish between acids and bases based on formula and chemical properties.
• Differentiate between concentration (molarity) and strength (degree of dissociation). No calculation involved.
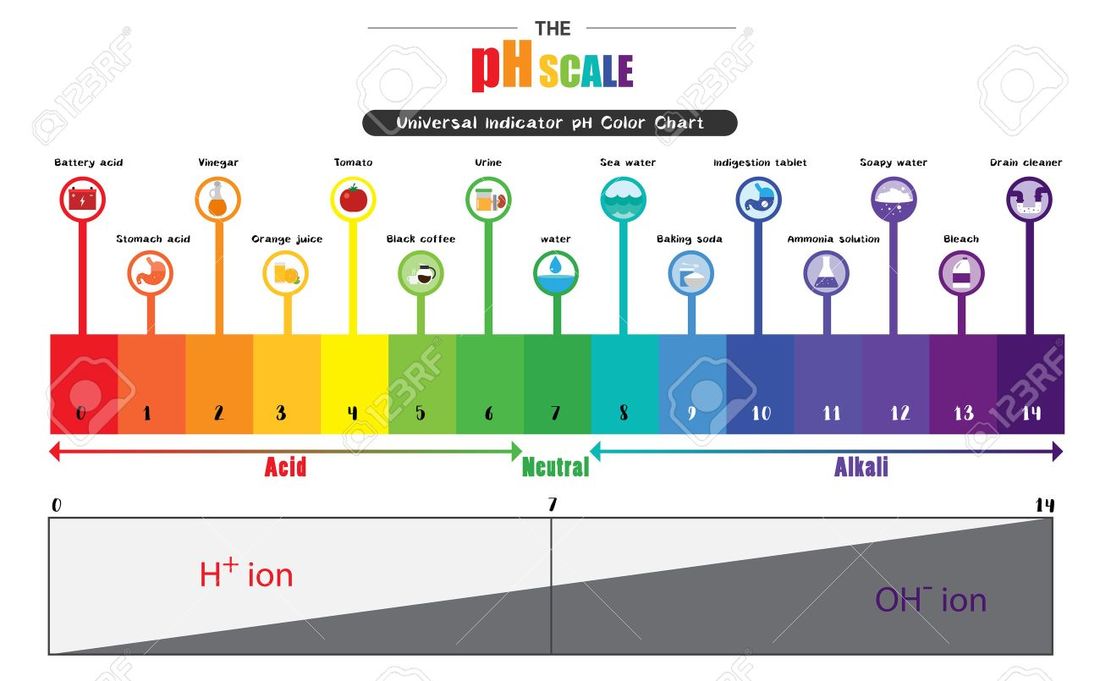
• Use pH scale to identify acids and bases.
• Interpret pH scale in terms of the exponential nature of pH values in terms of concentrations.
• Relate the color of indicator to pH using pH ranges provided in a table.
• Compute pH, pOH, [H+], and [OH-].
Chm.3.2.2 Summarize the properties of acids and bases.
● Distinguish properties of acids and bases related to taste, touch, reaction with metals, electrical conductivity, and identification with indicators such as litmus paper and phenolphthalein.
Chm.3.2.3 Infer the quantitative nature of a solution (molarity, dilution, and titration with a 1:1 molar ratio).
• Compute concentration (molarity) of solutions in moles per liter.
• Calculate molarity given mass of solute and volume of solution.
• Calculate mass of solute needed to create a solution of a given molarity and volume.
• Solve dilution problems: M1V1 = M2V2.
• Perform 1:1 titration calculations: MAVA = MBVB
• Determine the concentration of an acid or base using titration. Interpret titration curve for strong acid/strong base.
Chm.3.2.4 Summarize the properties of solutions.
• Identify types of solutions (solid, liquid, gaseous, aqueous).
• Define solutions as homogeneous mixtures in a single phase.
• Distinguish between electrolytic and nonelectrolytic solutions.
• Summarize colligative properties (vapor pressure reduction, boiling point elevation, freezing point depression, and osmotic pressure).
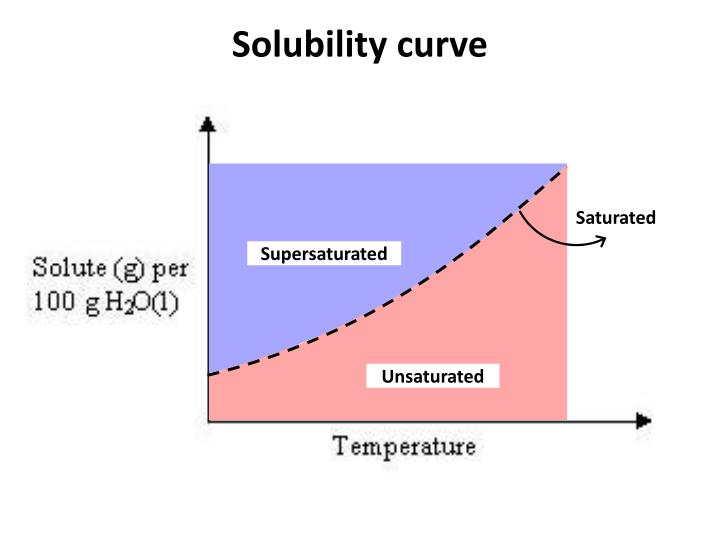
Chm.3.2.5 Interpret solubility diagrams.
• Use graph of solubility vs. temperature to identify a substance based on solubility at a particular temperature.
• Use graph to relate the degree of saturation of solutions to temperature.
• Develop a conceptual model for the solution process with a cause and effect relationship involving forces of attraction between solute
and solvent particles. A material is insoluble due to a lack of attraction between particles.
• Describe the energetics of the solution process as it occurs and the overall process as exothermic or endothermic.
• Explain solubility in terms of the nature of solute-solvent attraction, temperature and pressure (for gases).
Chm.3.2.1 Classify substances using the hydronium and hydroxide concentrations.
• Distinguish between acids and bases based on formula and chemical properties.
• Differentiate between concentration (molarity) and strength (degree of dissociation). No calculation involved.
• Use pH scale to identify acids and bases.
• Interpret pH scale in terms of the exponential nature of pH values in terms of concentrations.
• Relate the color of indicator to pH using pH ranges provided in a table.
• Compute pH, pOH, [H+], and [OH-].
Chm.3.2.2 Summarize the properties of acids and bases.
● Distinguish properties of acids and bases related to taste, touch, reaction with metals, electrical conductivity, and identification with indicators such as litmus paper and phenolphthalein.
Chm.3.2.3 Infer the quantitative nature of a solution (molarity, dilution, and titration with a 1:1 molar ratio).
• Compute concentration (molarity) of solutions in moles per liter.
• Calculate molarity given mass of solute and volume of solution.
• Calculate mass of solute needed to create a solution of a given molarity and volume.
• Solve dilution problems: M1V1 = M2V2.
• Perform 1:1 titration calculations: MAVA = MBVB
• Determine the concentration of an acid or base using titration. Interpret titration curve for strong acid/strong base.
Chm.3.2.4 Summarize the properties of solutions.
• Identify types of solutions (solid, liquid, gaseous, aqueous).
• Define solutions as homogeneous mixtures in a single phase.
• Distinguish between electrolytic and nonelectrolytic solutions.
• Summarize colligative properties (vapor pressure reduction, boiling point elevation, freezing point depression, and osmotic pressure).
Chm.3.2.5 Interpret solubility diagrams.
• Use graph of solubility vs. temperature to identify a substance based on solubility at a particular temperature.
• Use graph to relate the degree of saturation of solutions to temperature.
• Develop a conceptual model for the solution process with a cause and effect relationship involving forces of attraction between solute
and solvent particles. A material is insoluble due to a lack of attraction between particles.
• Describe the energetics of the solution process as it occurs and the overall process as exothermic or endothermic.
• Explain solubility in terms of the nature of solute-solvent attraction, temperature and pressure (for gases).
Vocabulary
https://www.123rf.com/photo_81803308_stock-vector-the-ph-scale-universal-indicator-ph-color-chart-diagram-acidic-alkaline-values-common-substances-vec.html
Acids & Bases
Learning Target(s)
I can distinguish between acids and bases.
Khan Academy: pH, pOH, and pH Scale
Calculating pH and pOH
Titration
Learning Target(s)
I can determine the concentration of an acid or base using titration.
http://springofchemistry.blogspot.com/2013/03/diagram-for-titration.html
Properties of Solutions
Learning Target(s)
I can interpret a solubility diagram
I know the difference between an unsaturated, saturated, and super saturated solution.
I know the difference between an unsaturated, saturated, and super saturated solution.
| |||||||