NC Standards
PSc.2.1 Understand types, properties, and structure of matter.
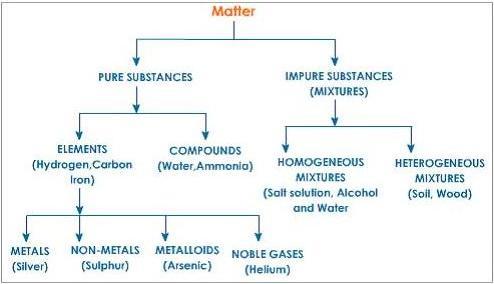
PSc.2.1.1 Classify matter as: homogeneous or heterogeneous; pure substance or mixture; element or compound; metals, nonmetals, or metalloids; solution, colloid, or suspension.
• Classify a sample of matter as homogeneous or heterogeneous based on uniformity of the material.
• Classify a sample of matter as a pure substance or mixture based on the number of elements or compounds in the sample.
• Classify an element as a metal, nonmetal, or metalloid based on its location on the periodic table.
• Classify a substance as an element or compound using its chemical formula.
• Classify samples and sets of matter as a solution, colloid or suspension based on the application of characteristic properties: particle size, “settling out” of one or more components, and interaction with light (Tyndall Effect).
PSc.2.1.2 Explain the phases of matter and the physical changes that matter undergoes.
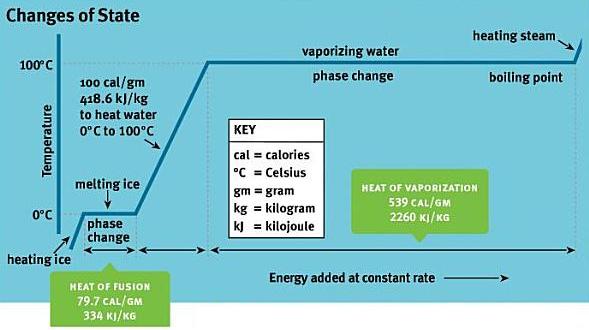
• Develop a conceptual cause-and-effect model for the phase change process that shows the relationship among particle attraction, particle motion, and gain or loss of heat - when a solid melts it has absorbed heat that increased the potential energy of its particles (space between particles) thus reducing the attraction between particles so that they can flow in a liquid phase. (Consider conditions of normal atmospheric pressure as well as the qualitative affects of changes in pressure involving gases.)
• The focus should be on the following phase changes: solid to liquid (melting), liquid to gas (vaporization), gas to liquid (condensation), and liquid to solid (freezing).
• Compare the process of evaporation to vaporization – materials that evaporate verses those which do not; attraction between surface particles and colliding air molecules.
• Recognize that the formation of solutions is a physical change forming a homogenous mixture. (Review from 8th grade.)
• Develop a conceptual model for the solution process with a cause and effect relationship involving forces of attraction between solute and solvent particles. A material is insoluble due to a lack of attraction between particles.
• Interpret solubility curves to determine the amount of solute that can dissolve in a given amount of solvent (typically water) at a given temperature.
• Qualitatively explain concentration of solutions as saturated, unsaturated or supersaturated; dilute or concentrated.
PSc.2.1.3 Compare physical and chemical properties of various types of matter.
• Calculate the density of different substances using the relationship D = m / V.
• Compare physical properties of a mixture that could be used to separate its components such as solubility, density, boiling point, magnetic property, etc.
• Compare various physical and chemical properties of metals, nonmetals and metalloids such as state of matter at a given temperature, density, melting point, boiling point, luster, conductivity, ductility, malleability, color, reactivity, etc.
• Compare physical and chemical properties of various everyday materials such as salt, sugar, baking soda, corn starch, rubbing alcohol, water, etc.
PSc.2.1.1 Classify matter as: homogeneous or heterogeneous; pure substance or mixture; element or compound; metals, nonmetals, or metalloids; solution, colloid, or suspension.
• Classify a sample of matter as homogeneous or heterogeneous based on uniformity of the material.
• Classify a sample of matter as a pure substance or mixture based on the number of elements or compounds in the sample.
• Classify an element as a metal, nonmetal, or metalloid based on its location on the periodic table.
• Classify a substance as an element or compound using its chemical formula.
• Classify samples and sets of matter as a solution, colloid or suspension based on the application of characteristic properties: particle size, “settling out” of one or more components, and interaction with light (Tyndall Effect).
PSc.2.1.2 Explain the phases of matter and the physical changes that matter undergoes.
• Develop a conceptual cause-and-effect model for the phase change process that shows the relationship among particle attraction, particle motion, and gain or loss of heat - when a solid melts it has absorbed heat that increased the potential energy of its particles (space between particles) thus reducing the attraction between particles so that they can flow in a liquid phase. (Consider conditions of normal atmospheric pressure as well as the qualitative affects of changes in pressure involving gases.)
• The focus should be on the following phase changes: solid to liquid (melting), liquid to gas (vaporization), gas to liquid (condensation), and liquid to solid (freezing).
• Compare the process of evaporation to vaporization – materials that evaporate verses those which do not; attraction between surface particles and colliding air molecules.
• Recognize that the formation of solutions is a physical change forming a homogenous mixture. (Review from 8th grade.)
• Develop a conceptual model for the solution process with a cause and effect relationship involving forces of attraction between solute and solvent particles. A material is insoluble due to a lack of attraction between particles.
• Interpret solubility curves to determine the amount of solute that can dissolve in a given amount of solvent (typically water) at a given temperature.
• Qualitatively explain concentration of solutions as saturated, unsaturated or supersaturated; dilute or concentrated.
PSc.2.1.3 Compare physical and chemical properties of various types of matter.
• Calculate the density of different substances using the relationship D = m / V.
• Compare physical properties of a mixture that could be used to separate its components such as solubility, density, boiling point, magnetic property, etc.
• Compare various physical and chemical properties of metals, nonmetals and metalloids such as state of matter at a given temperature, density, melting point, boiling point, luster, conductivity, ductility, malleability, color, reactivity, etc.
• Compare physical and chemical properties of various everyday materials such as salt, sugar, baking soda, corn starch, rubbing alcohol, water, etc.
Vocabulary
solid
liquid
gas
pure substance
element
compound
mixture
heterogeneous mixture
homogeneous mixture
suspension
solution
colloid
filtration
distillation
Charles' Law
Boyle's Law
phase change
endothermic
exothermic
vaporization
evaporation
vapor pressure
condensation
sublimation
deposition
liquid
gas
pure substance
element
compound
mixture
heterogeneous mixture
homogeneous mixture
suspension
solution
colloid
filtration
distillation
Charles' Law
Boyle's Law
phase change
endothermic
exothermic
vaporization
evaporation
vapor pressure
condensation
sublimation
deposition
Properties of Matter
Learning Targets
I can classify matter as homogeneous or heterogeneous, pure substance or mixture, element or compound, and solution, colloid, or suspension.
http://championapp.co/a-diagram-of-matter.html
| classifying_matter.pptx | |
| File Size: | 62 kb |
| File Type: | pptx |
Tyndall Effect
States of Matter
Learning Targets
I can compare physical and chemical properties of various types of matter.
https://chemistry.tutorvista.com/physical-chemistry/states-of-matter.html
| gas_law_and_phase_change.notebook | |
| File Size: | 119 kb |
| File Type: | notebook |
Transfer of Heat Energy
Learning Target(s)
I can explain thermal energy and its transfer.
I can predict the ability of materials to absorb or release thermal energy.
I can compare thermal energy, heat, and temperature.
I can describe how potential energy and kinetic energy are involved with phase changes.
I can compare conduction, convection, and radiation as methods of transferring heat.
I can predict the ability of materials to absorb or release thermal energy.
I can compare thermal energy, heat, and temperature.
I can describe how potential energy and kinetic energy are involved with phase changes.
I can compare conduction, convection, and radiation as methods of transferring heat.
| energy_conversion_and_conservation.ppt | |
| File Size: | 811 kb |
| File Type: | ppt |
| heat_and_temp.ppt | |
| File Size: | 1613 kb |
| File Type: | ppt |
Phase Changes
Learning Targets
I can explain the phases of matter and the physical changes that matter undergoes.
Phase Diagrams
Gas Laws
Learning Targets
I can explain the phases of matter and the physical changes that matter undergoes.