Which Periodic Element Are You?
Interactive Periodic Table
| activity-ptable-student-periodictable.docx | |
| File Size: | 44 kb |
| File Type: | docx |
Periodic Videos
NC Standards
Chm.1.3 Understand the physical and chemical properties of atoms based on their position on the Periodic Table.
Chm.1.3.1 Classify the components of a periodic table (period, group, metal, metalloid, nonmetal, transition).
Using the Periodic Table,
Groups (families)
Identify groups as vertical columns on the periodic table.
Know that main group elements in the same group have similar properties, the same number of valence electrons, and the same oxidation number.
Summarize that reactivity increases as you go down within a group for metals and decreases for nonmetals.
Periods
Identify periods as horizontal rows on the periodic table. Metals/Nonmetals/Metalloids Identify regions of the periodic table where metals, nonmetals, and metalloids are located.
Classify elements as metals/nonmetals/metalloids based on location. Representative elements (main group) and transition elements.
Identify representative (main group) elements as A groups or as groups 1, 2, 13-18.
Identify alkali metals, alkaline earth metals, halogens, and noble gases based on location on periodic table.
Identify transition elements as B groups or as groups 3-12.
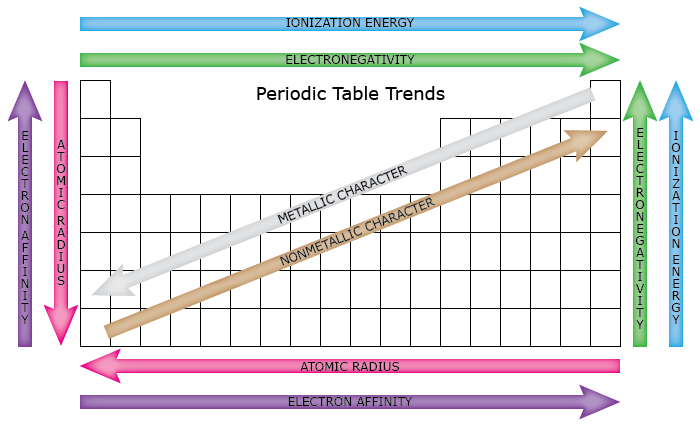
Chm.1.3.2 Infer the physical properties (atomic radius, metallic and nonmetallic characteristics) of an element based on its position on the Periodic Table.
Using the Periodic Table,
Define atomic radius and ionic radius.
Know group and period general trends for atomic radius.
Apply trends to arrange elements in order of increasing or decreasing atomic radius. Explain the reasoning behind the trends. Compare cation and anion radius to neutral atom.
Write electron configurations, including noble gas abbreviations (no exceptions to the general rules). Included here are extended arrangements showing electrons in orbitals.
Identify s, p, d, and f blocks on Periodic Table.
Identify an element based on its electron configuration. (Students should be able to identify elements which follow the general rules, not necessarily those which are exceptions.)
Determine the number of valence electrons from electron configurations.
Compare the metallic character of elements.
Use electron configuration and ion formation to justify metallic character. (Metals tend to lose electrons in order to achieve the stability of a filled octet.)
Relate metallic character to ionization energy and electronegativity.
Chm.1.3.3 Infer the atomic size, reactivity, electronegativity, and ionization energy of an element from its position on the Periodic Table.
Using the Periodic Table,
Predict the number of electrons lost or gained and the oxidation number based on the electron configuration of an atom.
Explain how the general size of an atom contributes to its reactivity‒sharing, gaining or losing electrons.
Compare reactivity of elements within groups and periods of the periodic table.
Define ionization energy and know group and period general trends for ionization energy. Explain the reasoning behind the trend.
Apply trends to arrange elements in order of increasing or decreasing ionization energy.
Define electronegativity and know group and period general trends for electronegativity. Explain the reasoning behind the trend. Apply trends to arrange elements in order of increasing or decreasing electronegativity.
Chm.1.3.1 Classify the components of a periodic table (period, group, metal, metalloid, nonmetal, transition).
Using the Periodic Table,
Groups (families)
Identify groups as vertical columns on the periodic table.
Know that main group elements in the same group have similar properties, the same number of valence electrons, and the same oxidation number.
Summarize that reactivity increases as you go down within a group for metals and decreases for nonmetals.
Periods
Identify periods as horizontal rows on the periodic table. Metals/Nonmetals/Metalloids Identify regions of the periodic table where metals, nonmetals, and metalloids are located.
Classify elements as metals/nonmetals/metalloids based on location. Representative elements (main group) and transition elements.
Identify representative (main group) elements as A groups or as groups 1, 2, 13-18.
Identify alkali metals, alkaline earth metals, halogens, and noble gases based on location on periodic table.
Identify transition elements as B groups or as groups 3-12.
Chm.1.3.2 Infer the physical properties (atomic radius, metallic and nonmetallic characteristics) of an element based on its position on the Periodic Table.
Using the Periodic Table,
Define atomic radius and ionic radius.
Know group and period general trends for atomic radius.
Apply trends to arrange elements in order of increasing or decreasing atomic radius. Explain the reasoning behind the trends. Compare cation and anion radius to neutral atom.
Write electron configurations, including noble gas abbreviations (no exceptions to the general rules). Included here are extended arrangements showing electrons in orbitals.
Identify s, p, d, and f blocks on Periodic Table.
Identify an element based on its electron configuration. (Students should be able to identify elements which follow the general rules, not necessarily those which are exceptions.)
Determine the number of valence electrons from electron configurations.
Compare the metallic character of elements.
Use electron configuration and ion formation to justify metallic character. (Metals tend to lose electrons in order to achieve the stability of a filled octet.)
Relate metallic character to ionization energy and electronegativity.
Chm.1.3.3 Infer the atomic size, reactivity, electronegativity, and ionization energy of an element from its position on the Periodic Table.
Using the Periodic Table,
Predict the number of electrons lost or gained and the oxidation number based on the electron configuration of an atom.
Explain how the general size of an atom contributes to its reactivity‒sharing, gaining or losing electrons.
Compare reactivity of elements within groups and periods of the periodic table.
Define ionization energy and know group and period general trends for ionization energy. Explain the reasoning behind the trend.
Apply trends to arrange elements in order of increasing or decreasing ionization energy.
Define electronegativity and know group and period general trends for electronegativity. Explain the reasoning behind the trend. Apply trends to arrange elements in order of increasing or decreasing electronegativity.
Vocabulary
group/family
column
valence electrons
oxidation number
reactivity (trend for metals and nonmetals)
rows
periods
metals
nonmetals
metalloids
alkali metals
alkaline earth metals
halogens
noble gases
A groups (representative elements)
B groups (transition elements)
atomic radius
ionic radius (cation and anion)
metallic character
atomic size
ionization energy
electron affinity
electronegativity
electron configurations
column
valence electrons
oxidation number
reactivity (trend for metals and nonmetals)
rows
periods
metals
nonmetals
metalloids
alkali metals
alkaline earth metals
halogens
noble gases
A groups (representative elements)
B groups (transition elements)
atomic radius
ionic radius (cation and anion)
metallic character
atomic size
ionization energy
electron affinity
electronegativity
electron configurations
Review Games
Choice Board
| periodic_trends_pages_1_4.doc | |
| File Size: | 71 kb |
| File Type: | doc |
| periodictabletrendsworksheet.doc | |
| File Size: | 36 kb |
| File Type: | doc |
| electronegativtyws1.pdf | |
| File Size: | 25 kb |
| File Type: | |
| electronegativityws2.pdf | |
| File Size: | 35 kb |
| File Type: | |
| periodic_table_project.doc | |
| File Size: | 30 kb |
| File Type: | doc |
Components
Learning Targets
I can classify the components of a periodic table.
| hist_periodictable.ppt | |
| File Size: | 616 kb |
| File Type: | ppt |
| periodic_table_on_the_internetname_page_1.doc | |
| File Size: | 31 kb |
| File Type: | doc |
| blank_periodic_table.docx | |
| File Size: | 118 kb |
| File Type: | docx |
Metals, Nonmetals, and Metalloids of the Periodic Table?
Element Families of the Periodic Table
Properties
Learning Targets
I can use the periodic table to predict atomic and ionic radii, metallic and nonmetallic character.
I can use the periodic table to predict atomic size, electron configurations, ionization energy, electronegativity, and electron affinity.
I can use the periodic table to predict atomic size, electron configurations, ionization energy, electronegativity, and electron affinity.
| periodic_trends_pres.ppt | |
| File Size: | 349 kb |
| File Type: | ppt |
| unit_2_-_periodic_trends_ol.pptx | |
| File Size: | 379 kb |
| File Type: | pptx |
| unit_2_periodic_trends.pptx | |
| File Size: | 932 kb |
| File Type: | pptx |
Periodic Trends Game
Science Geek: Periodic Trends
https://sciencenotes.org/periodic-table-trends/
Oxidation Numbers
Learning Targets
I can interpret the oxidation rules.
I can use the periodic table to predict oxidation numbers for elements.
I can use the periodic table to predict oxidation numbers for elements.