NC Standards
Chm.2.1 Understand the relationship among pressure, temperature, volume, and phase.
Chm.2.1.1 Explain the energetic nature of phase changes.
• Explain physical equilibrium: liquid water-water vapor. Vapor pressure depends on temperature and concentration of particles in solution. (conceptual only – no calculations)
• Explain how the energy (kinetic and potential) of the particles of a substance changes when heated, cooled, or changing phase.
• Identify pressure as well as temperature as a determining factor for phase of matter.
• Contrast heat and temperature, including temperature as a measure of average kinetic energy, and appropriately use the units Joule, Celsius, and Kelvin.
Chm.2.1.2 Explain heating and cooling curves (heat of fusion, heat of vaporization, heat, melting point, and boiling point).
• Define and use the terms and/or symbols for: specific heat capacity, heat of fusion, heat of vaporization.
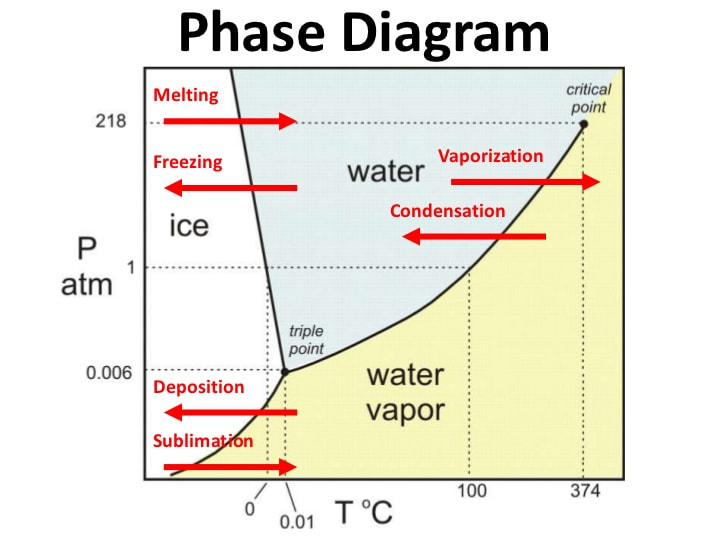
• Interpret the following: heating and cooling curves (noting both significance of plateaus and the physical states of each segment. Phase diagrams for H2O and CO2.
• Complete calculations of: q=mCp∆T, q = mHf , and q = mHv using heatling/cooling curve data.
• Explain phase change calculations in terms of heat absorbed or released (endothermic vs. exothermic processes).
Chm.2.1.3 Interpret the data presented in phase diagrams.
• Draw phase diagrams of water and carbon dioxide (shows how sublimation occurs). Identify regions, phases and phase changes using a phase diagram.
• Use phase diagrams to determine information such as (1) phase at a given temperature and pressure, (2) boiling point or melting point at a given pressure, (3) triple point of a material.
Chm.2.1.4 Infer simple calorimetric calculations based on the concepts of heat lost equals heat gained and specific heat.
• Recognize that, for a closed system, energy is neither lost nor gained only transferred between components of the system.
• Complete calculations of: q=mCp∆T, q = mHf , q = mHv, and q lost=(-q gain) in water, including phase changes, using laboratory data.
Chm.2.1.1 Explain the energetic nature of phase changes.
• Explain physical equilibrium: liquid water-water vapor. Vapor pressure depends on temperature and concentration of particles in solution. (conceptual only – no calculations)
• Explain how the energy (kinetic and potential) of the particles of a substance changes when heated, cooled, or changing phase.
• Identify pressure as well as temperature as a determining factor for phase of matter.
• Contrast heat and temperature, including temperature as a measure of average kinetic energy, and appropriately use the units Joule, Celsius, and Kelvin.
Chm.2.1.2 Explain heating and cooling curves (heat of fusion, heat of vaporization, heat, melting point, and boiling point).
• Define and use the terms and/or symbols for: specific heat capacity, heat of fusion, heat of vaporization.
• Interpret the following: heating and cooling curves (noting both significance of plateaus and the physical states of each segment. Phase diagrams for H2O and CO2.
• Complete calculations of: q=mCp∆T, q = mHf , and q = mHv using heatling/cooling curve data.
• Explain phase change calculations in terms of heat absorbed or released (endothermic vs. exothermic processes).
Chm.2.1.3 Interpret the data presented in phase diagrams.
• Draw phase diagrams of water and carbon dioxide (shows how sublimation occurs). Identify regions, phases and phase changes using a phase diagram.
• Use phase diagrams to determine information such as (1) phase at a given temperature and pressure, (2) boiling point or melting point at a given pressure, (3) triple point of a material.
Chm.2.1.4 Infer simple calorimetric calculations based on the concepts of heat lost equals heat gained and specific heat.
• Recognize that, for a closed system, energy is neither lost nor gained only transferred between components of the system.
• Complete calculations of: q=mCp∆T, q = mHf , q = mHv, and q lost=(-q gain) in water, including phase changes, using laboratory data.
Vocabulary
chemical change
liquid
chemical property
mass
chemical reaction
mixture
chemical symbol
phase
compound
physical change
distillation
physical property
element
precipitate
extensive property
product
filtration
reactant
gas
solid
heterogeneous mixture
solution
homogeneous mixture
substance
intensive property
vapor
volume
specific heatatmospheric pressure
barometer
boiling point
crystal
evaporation
gas pressure
glass
kinetic energy
kinetic theory
melting point
normal boiling point
pascal (Pa)
phase diagram
standard atmosphere (atm)
sublimation
triple point
unit cell
vacuum
vaporization
vapor pressure
liquid
chemical property
mass
chemical reaction
mixture
chemical symbol
phase
compound
physical change
distillation
physical property
element
precipitate
extensive property
product
filtration
reactant
gas
solid
heterogeneous mixture
solution
homogeneous mixture
substance
intensive property
vapor
volume
specific heatatmospheric pressure
barometer
boiling point
crystal
evaporation
gas pressure
glass
kinetic energy
kinetic theory
melting point
normal boiling point
pascal (Pa)
phase diagram
standard atmosphere (atm)
sublimation
triple point
unit cell
vacuum
vaporization
vapor pressure
Matter and Change
Learning Targets
I can analyze the evidence of chemical change.
I can identify types of solutions.
I can identify types of solutions.
| chem_matter_and_change-4.pptx | |
| File Size: | 63 kb |
| File Type: | pptx |
Kinetic Theory and States of Matter
Learning Targets
I can explain the energetic nature of phases.
I can explain how the energy (kinetic and potential) of the particles of a substance changes when heated, cooled, or changing phases.
I can explain how the energy (kinetic and potential) of the particles of a substance changes when heated, cooled, or changing phases.
Phase Diagrams
Learning Targets
I can interpret the data presented in phase diagrams.
| phase_diagram_worksheet.docx | |
| File Size: | 35 kb |
| File Type: | docx |
https://www.slideshare.net/joverba/phase-diagram-notes
Thermochemistry
Learning Targets
I can infer simple calorimetric calculation based on the concepts of of heat lost equals heat gained and specific heat.
| chem_jan_7.pptx | |
| File Size: | 85 kb |
| File Type: | pptx |
| chem_sp_heat_homework.docx | |
| File Size: | 15 kb |
| File Type: | docx |