NC Standards
PSc.2.2 Understand chemical bonding and chemical interactions.
PSc.2.2.2 Infer the type of chemical bond that occurs, whether covalent, ionic, or metallic, in a given substance.
• Describe how ionic, covalent, and metallic bonds form and provide examples of substances that exhibit each type of bonding.
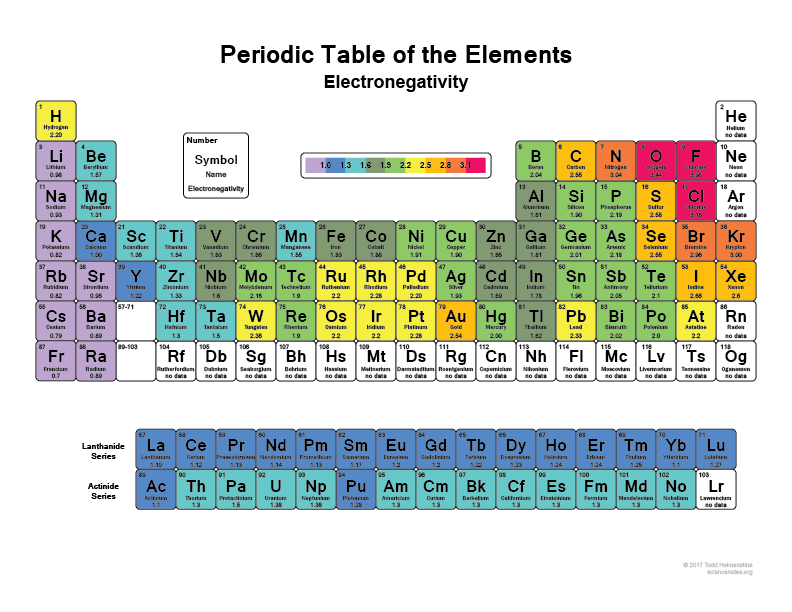
• Predict the type of bond between two elements in a compound based on their positions in the periodic table.
PSc.2.2 Understand chemical bonding and chemical interactions.
PSc.2.2.3 Predict chemical formulas and names for simple compounds based on knowledge of bond formation and naming conventions.
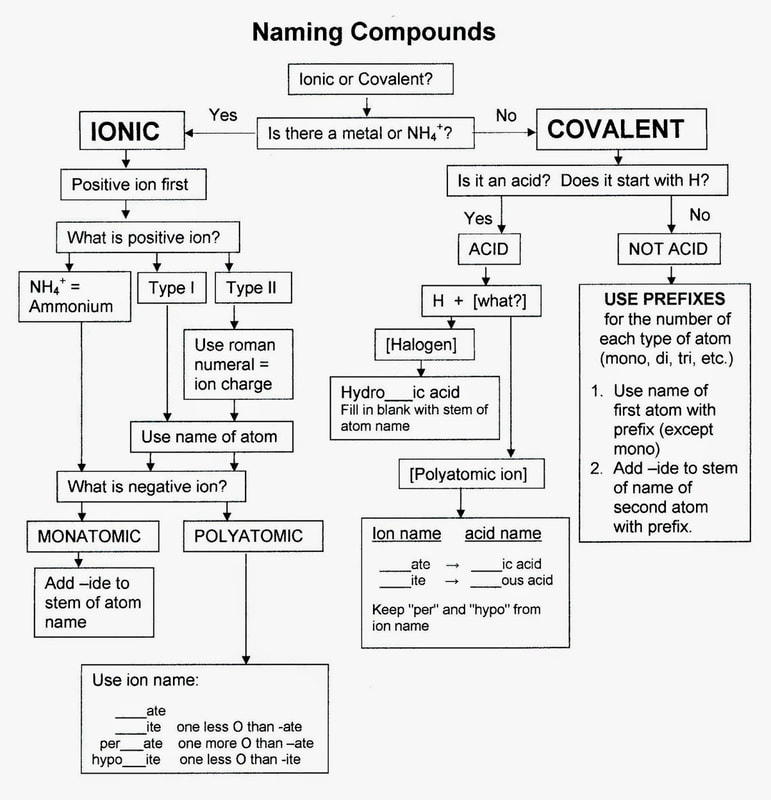
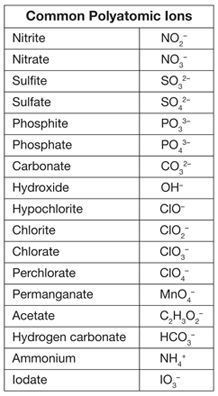
• Name and write formulas for simple binary compounds containing a metal and nonmetal using representative elements (A Groups or 1, 2, 13-18) and compounds involving common polyatomic ions: ammonium (NH4⁺), acetate (C2H3O2⁻), nitrate (NO3⁻), hydroxide (OH⁻), carbonate (CO32⁻), sulfate (SO42⁻), phosphate (PO43⁻).
• Name and write formulas for binary compounds of two nonmetals using Greek prefixes (mono-, di-, tri-, tetra-, etc.).
PSc.2.2.2 Infer the type of chemical bond that occurs, whether covalent, ionic, or metallic, in a given substance.
• Describe how ionic, covalent, and metallic bonds form and provide examples of substances that exhibit each type of bonding.
• Predict the type of bond between two elements in a compound based on their positions in the periodic table.
PSc.2.2 Understand chemical bonding and chemical interactions.
PSc.2.2.3 Predict chemical formulas and names for simple compounds based on knowledge of bond formation and naming conventions.
• Name and write formulas for simple binary compounds containing a metal and nonmetal using representative elements (A Groups or 1, 2, 13-18) and compounds involving common polyatomic ions: ammonium (NH4⁺), acetate (C2H3O2⁻), nitrate (NO3⁻), hydroxide (OH⁻), carbonate (CO32⁻), sulfate (SO42⁻), phosphate (PO43⁻).
• Name and write formulas for binary compounds of two nonmetals using Greek prefixes (mono-, di-, tri-, tetra-, etc.).
Vocabulary
chemical bond
ionic bond
chemical formula
crystals
covalent bond
molecule
polar covalent bond
metallic bond
alloy
ion
anion
cation
ionic bond
chemical formula
crystals
covalent bond
molecule
polar covalent bond
metallic bond
alloy
ion
anion
cation
https://lavelle.chem.ucla.edu/forum/viewtopic.php?t=15381
Ionic Bonding
Learning Targets
I can describe how ionic bonds form.
I can predict what bond will form between two elements based on their positions in the periodic table.
I can construct ionic compounds.
I can name ionic compounds using IUPAC convention.
I can predict what bond will form between two elements based on their positions in the periodic table.
I can construct ionic compounds.
I can name ionic compounds using IUPAC convention.
| ps_ionic_bonding.pptx | |
| File Size: | 311 kb |
| File Type: | pptx |
https://www.pinterest.com/pin/244601823489877457/
Covalent Bonding
Learning Targets
I can describe how covalent bonds form.
I can predict what bond will form between two elements based on their positions in the periodic table.
I can construct formulas for molecular compounds.
I can name molecular compounds using IUPAC convention.
I can predict what bond will form between two elements based on their positions in the periodic table.
I can construct formulas for molecular compounds.
I can name molecular compounds using IUPAC convention.
| pscovalent_bonding.pptx | |
| File Size: | 199 kb |
| File Type: | pptx |
Electronegativity
Polyatomic Ions
Learning Targets
I can write formulas for ionic compounds with polyatomic ions.
I can name formulas with polyatomic ions using IUPAC convention.
I can name formulas with polyatomic ions using IUPAC convention.
https://www.pinterest.com/pin/863706034757076244/
Metallic Bonding
I can describe bow metallic bonds form.
| ps_metallic_bonding.pptx | |
| File Size: | 70 kb |
| File Type: | pptx |