NC Standards
PSc.2.2 Understand chemical bonding and chemical interactions.
PSc.2.2.6
•Recognize common inorganic acids including hydrochloric (muriatic) acid, sulfuric acid, acetic acid, nitric acid and citric acid.
•Recognize common bases including sodium bicarbonate, and hydroxides of sodium, potassium, calcium, magnesium, barium and
ammonium.
•Define acids and bases according to the Arrhenius theory.
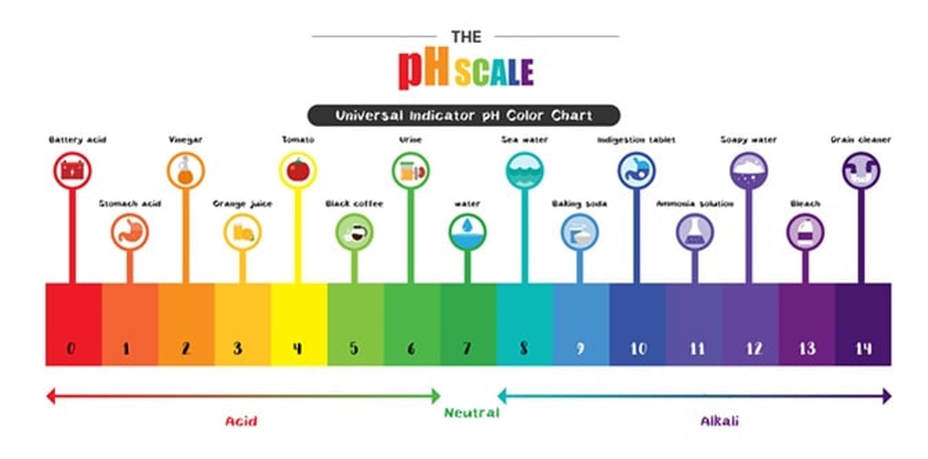
•Develop an understanding of the pH scale and the classification of substances therein.
•Generalize common characteristics of acids and bases– pH range, reactivity with metals and carbonates (acids) or fats/oils (bases),
conductivity.
•Relate general household uses of acids and bases with their characteristic properties.
•Explain what happens in a neutralization reaction, identifying each component substance.
PSc.2.2.6
•Recognize common inorganic acids including hydrochloric (muriatic) acid, sulfuric acid, acetic acid, nitric acid and citric acid.
•Recognize common bases including sodium bicarbonate, and hydroxides of sodium, potassium, calcium, magnesium, barium and
ammonium.
•Define acids and bases according to the Arrhenius theory.
•Develop an understanding of the pH scale and the classification of substances therein.
•Generalize common characteristics of acids and bases– pH range, reactivity with metals and carbonates (acids) or fats/oils (bases),
conductivity.
•Relate general household uses of acids and bases with their characteristic properties.
•Explain what happens in a neutralization reaction, identifying each component substance.
Vocabulary
Acids
Bases
Arrhenius Theory
Brownsted-Lowry Theory
Concentrated Solution
Corrosive
Dilute Solution
Hydrogen Ion
Hydroxide Ion
Indicator
Neutralization
pH scale
Salt
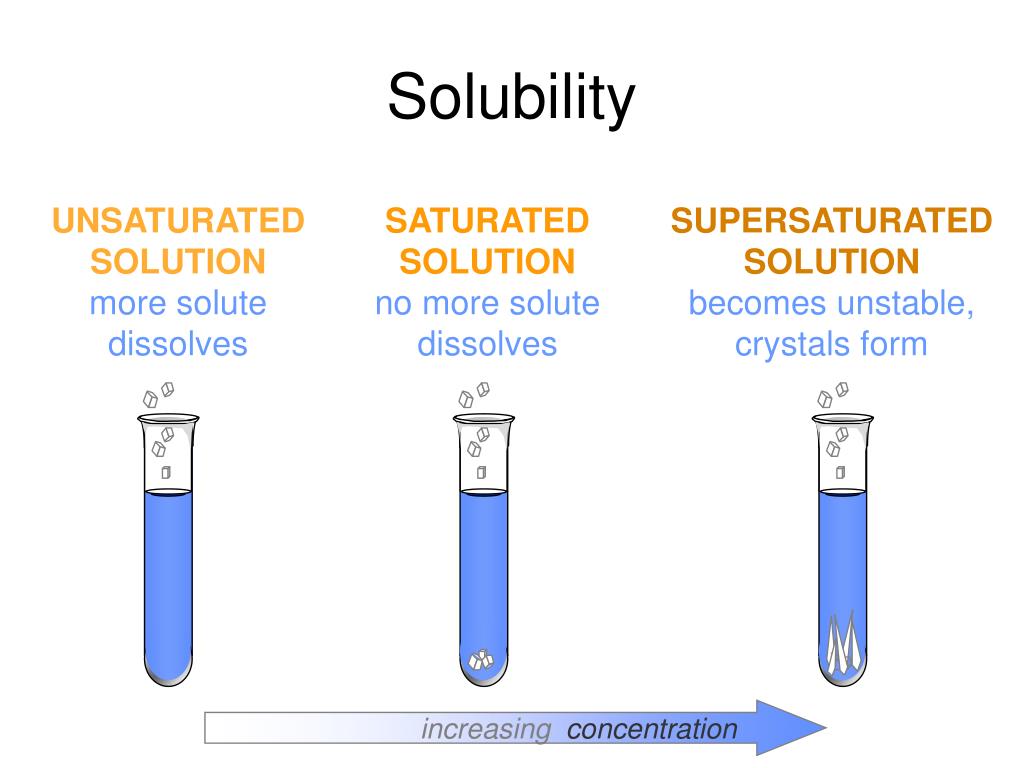
Saturated Solution
Solute
Solvent
Solubility
Supersaturated Solution
Unsaturated Solution
Bases
Arrhenius Theory
Brownsted-Lowry Theory
Concentrated Solution
Corrosive
Dilute Solution
Hydrogen Ion
Hydroxide Ion
Indicator
Neutralization
pH scale
Salt
Saturated Solution
Solute
Solvent
Solubility
Supersaturated Solution
Unsaturated Solution
Quizlet: Physical Science-Acids, Bases, and Solutions
Acids vs. Bases
Learning Target(s)
I can recognize common acids and bases by their characteristics.
I can define acids and bases by the Arrhenius theory.
I can can interpret and create a pH scale and properly place items based on their pH's.
I can define acids and bases by the Arrhenius theory.
I can can interpret and create a pH scale and properly place items based on their pH's.
The Nature of Acids and Bases
https://www.news-medical.net/health/pH-in-the-Human-Body.aspx
Nomenclature
Learning Target(s)
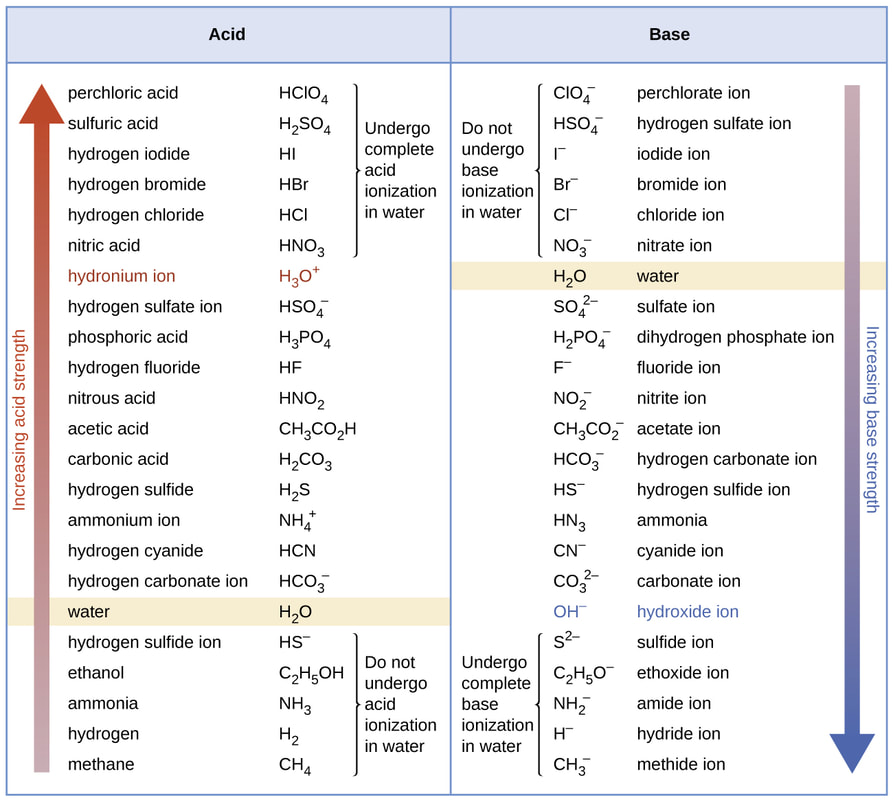
I can identify and name acids and bases using their chemical formula and vise versa.
Naming Acids and Bases
https://opentextbc.ca/chemistry/chapter/14-3-relative-strengths-of-acids-and-bases/
Solutions
Learning Target(s)
Types of Saturation
Saturated and Unsaturated Solutions
https://www.slideserve.com/trula/solubility