NC Standards
Chm.1.1 Analyze the structure of atoms and ions.
Chm.1.1.1 Analyze the structure of atoms, isotopes, and ions.
Characterize protons, neutrons, electrons by location, relative charge, relative mass (p=1, n=1, e=1/2000).
Use symbols: A= mass number, Z=atomic number
Use notation for writing isotope symbols: 235 U or U-23592
Identify isotope using mass number and atomic number and relate to number of protons, neutrons and electrons.
Differentiate average atomic mass of an element from the actual isotopic mass and mass number of specific isotopes.
Use example calculations to determine average atomic mass of atoms from relative abundance and actual isotopic mass to develop understanding.
Chm.1.1.2 Analyze an atom in terms of the location of electrons.
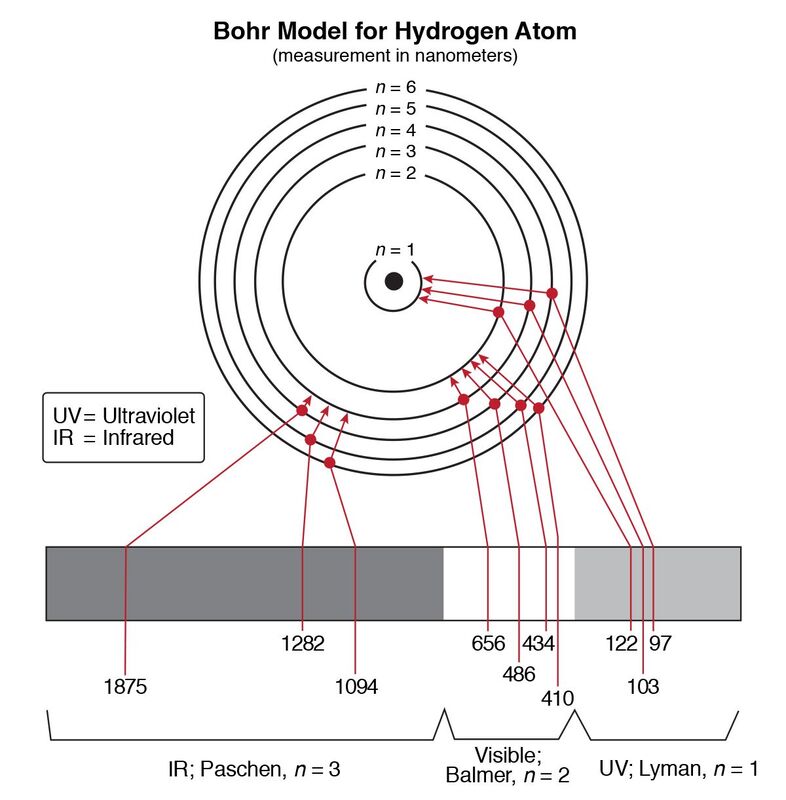
Analyze diagrams related to the Bohr model of the hydrogen atom in terms of allowed, discrete energy levels in the emission spectrum. Describe the electron cloud of the atom in terms of a probability model.
Relate the electron configurations of atoms to the Bohr and electron cloud models.
Chm.1.1.3 Explain the emission of electromagnetic radiation in spectral form in terms of the Bohr model.
Understand that energy exists in discrete units called quanta.
Describe the concepts of excited and ground state of electrons in the atom: 1. When an electron gains an amount of energy equivalent to the energy difference, it moves from its ground state to a higher
energy level.
When the electron moves to a lower energy level, it releases an amount of energy equal to the energy difference in these levels as electromagnetic radiation (emissions spectrum).
Articulate that this electromagnetic radiation is given off as photons.
Understand the inverse relationship between wavelength and frequency, and the direct relationship between energy and frequency.
Use the “Bohr Model for Hydrogen Atom” and “Electromagnetic Spectrum” diagrams from the Reference Tables to relate color,
frequency, and wavelength of the light emitted to the energy of the photon.
Explain that Niels Bohr produced a model of the hydrogen atom based on experimental observations.
This model indicated that: 1. an electron circles the nucleus only in fixed energy ranges called orbits; 2. an electron can neither gain or lose energy inside this orbit, but could move up or down to another orbit; 3. that the lowest energy orbit is closest to the nucleus.
Describe the wave/particle duality of electrons.
Chm.1.1.4 Explain the process of radioactive decay by the use of nuclear equations and half-life.
Use the symbols for and distinguish between alpha ( 24He), and beta ( -10e) nuclear particles, and gamma radiation include relative mass.
Use shorthand notation of particles involved in nuclear equations to balance and solve for unknowns.
Compare the penetrating ability of alpha, beta, and gamma radiation.
Conceptually describe nuclear decay, including
1. Decay as a random event, independent of other energy influences
2. Using symbols to represent simple balanced decay equation
3. Half-life (including simple calculations).
Compare radioactive decay with fission and fusion.
Chm.1.1.1 Analyze the structure of atoms, isotopes, and ions.
Characterize protons, neutrons, electrons by location, relative charge, relative mass (p=1, n=1, e=1/2000).
Use symbols: A= mass number, Z=atomic number
Use notation for writing isotope symbols: 235 U or U-23592
Identify isotope using mass number and atomic number and relate to number of protons, neutrons and electrons.
Differentiate average atomic mass of an element from the actual isotopic mass and mass number of specific isotopes.
Use example calculations to determine average atomic mass of atoms from relative abundance and actual isotopic mass to develop understanding.
Chm.1.1.2 Analyze an atom in terms of the location of electrons.
Analyze diagrams related to the Bohr model of the hydrogen atom in terms of allowed, discrete energy levels in the emission spectrum. Describe the electron cloud of the atom in terms of a probability model.
Relate the electron configurations of atoms to the Bohr and electron cloud models.
Chm.1.1.3 Explain the emission of electromagnetic radiation in spectral form in terms of the Bohr model.
Understand that energy exists in discrete units called quanta.
Describe the concepts of excited and ground state of electrons in the atom: 1. When an electron gains an amount of energy equivalent to the energy difference, it moves from its ground state to a higher
energy level.
When the electron moves to a lower energy level, it releases an amount of energy equal to the energy difference in these levels as electromagnetic radiation (emissions spectrum).
Articulate that this electromagnetic radiation is given off as photons.
Understand the inverse relationship between wavelength and frequency, and the direct relationship between energy and frequency.
Use the “Bohr Model for Hydrogen Atom” and “Electromagnetic Spectrum” diagrams from the Reference Tables to relate color,
frequency, and wavelength of the light emitted to the energy of the photon.
Explain that Niels Bohr produced a model of the hydrogen atom based on experimental observations.
This model indicated that: 1. an electron circles the nucleus only in fixed energy ranges called orbits; 2. an electron can neither gain or lose energy inside this orbit, but could move up or down to another orbit; 3. that the lowest energy orbit is closest to the nucleus.
Describe the wave/particle duality of electrons.
Chm.1.1.4 Explain the process of radioactive decay by the use of nuclear equations and half-life.
Use the symbols for and distinguish between alpha ( 24He), and beta ( -10e) nuclear particles, and gamma radiation include relative mass.
Use shorthand notation of particles involved in nuclear equations to balance and solve for unknowns.
Compare the penetrating ability of alpha, beta, and gamma radiation.
Conceptually describe nuclear decay, including
1. Decay as a random event, independent of other energy influences
2. Using symbols to represent simple balanced decay equation
3. Half-life (including simple calculations).
Compare radioactive decay with fission and fusion.
Vocabulary
quanta
excited state
ground state
energy
emission spectra
electromagnetic radiation (spectra)
wavelength frequency
color of light
photon
orbits
wave/particle duality
quantum numbers
sublevels (s, p, d, f)
excited state
ground state
energy
emission spectra
electromagnetic radiation (spectra)
wavelength frequency
color of light
photon
orbits
wave/particle duality
quantum numbers
sublevels (s, p, d, f)
Isotopes
Learning Targets
I can identify different isotopes.
I can use different isotopes and their percentages to calculate average atomic mass of elements.
I can use different isotopes and their percentages to calculate average atomic mass of elements.
Isotopes and Calculating Average Atomic Mass
https://teachchemistry.org/periodical/issues/may-2017/isotopes-calculating-average-atomic-mass
teachchemistry.org/isotopes
teachchemistry.org/isotopes
Bohr Model
Learning Targets
I can describe the relationship between an electron’s movement and its energy.
I can relate the energy produced to the type of radiation produced in the electromagnetic spectrum.
I can describe the role of experimental observation in constructing models of the atom (Bohr and de Broglie)
I can relate the energy produced to the type of radiation produced in the electromagnetic spectrum.
I can describe the role of experimental observation in constructing models of the atom (Bohr and de Broglie)
| the_bohr_atom.ppt | |
| File Size: | 295 kb |
| File Type: | ppt |
| bohrandlewis-4.ppt | |
| File Size: | 270 kb |
| File Type: | ppt |
| bohr_model_worksheet.docx | |
| File Size: | 15 kb |
| File Type: | docx |
| electon_lab.doc | |
| File Size: | 34 kb |
| File Type: | doc |
Line Spectra and the Bohr Model
Learning Targets
I can explain when the electron moves to a lower energy level, it releases an amount of energy equal to the energy difference in these levels as electromagnetic radiation (emissions spectrum)
I can use the “Bohr Model for Hydrogen Atom” and “Electromagnetic Spectrum” diagrams from the Reference Tables to relate color,
frequency, and wavelength of the light emitted to the energy of the photon.
I can use the “Bohr Model for Hydrogen Atom” and “Electromagnetic Spectrum” diagrams from the Reference Tables to relate color,
frequency, and wavelength of the light emitted to the energy of the photon.
LibreTexts Line Spectra and the Bohr Model
Electron Configuration
Learning Targets
I can describe the location of an electron based on the Bohr model and the quantum theory.
LibreTexts Electron Configurations
| quantum_model_of_the_atom.ppt | |
| File Size: | 2548 kb |
| File Type: | ppt |
| electron_config_graphic_organizer.docx | |
| File Size: | 146 kb |
| File Type: | docx |