NC Standards
PSc.2.1.4
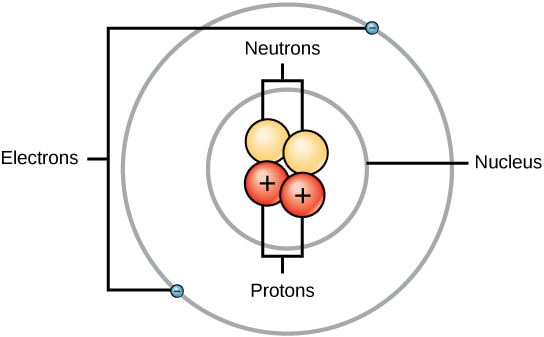
• Describe the charge, relative mass, and the location of protons, electrons, and neutrons within an atom.
• Calculate the number of protons, neutrons, electrons, and mass number in neutral atoms and ions.
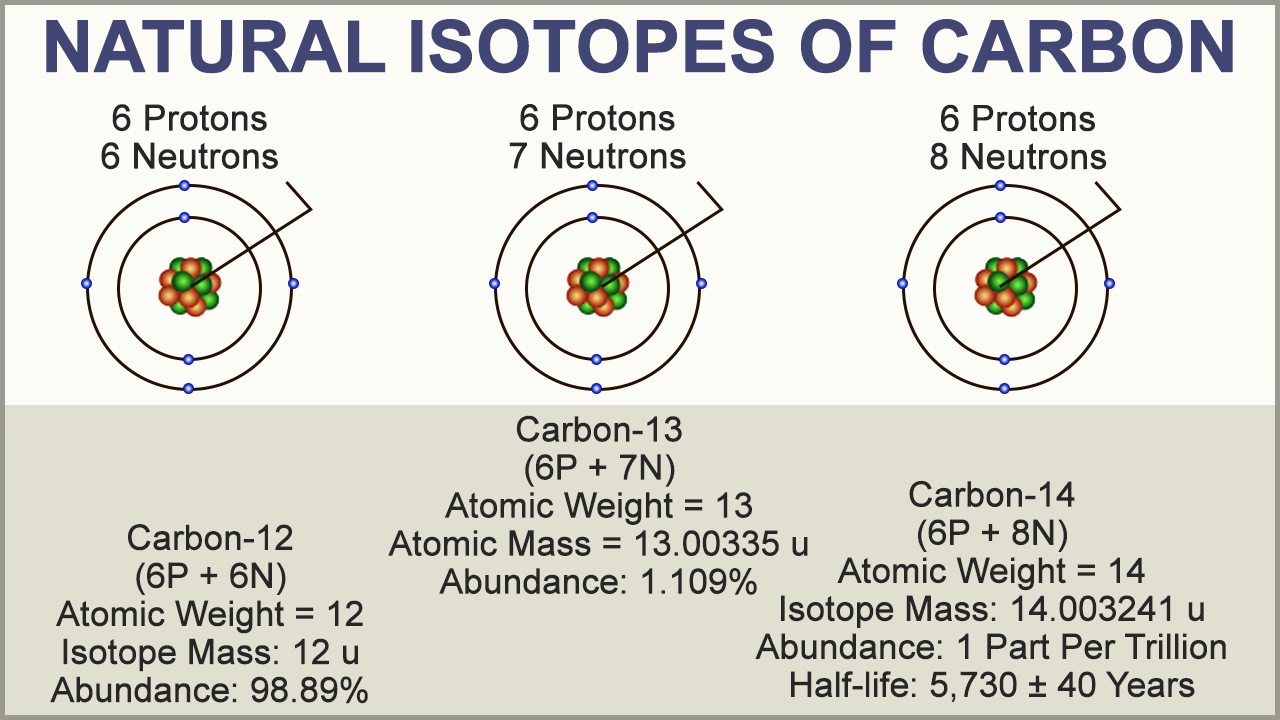
• Explain how the different mass numbers of isotopes contributes to the average atomic mass for a given element (conceptual, no
calculations).
• Use isotopic notation to write symbols for various isotopes (ex. Carbon-12, C-12, 12C, etc.)
• Explain Bohr’s model of the atom.
• Draw Bohr models from hydrogen to argon including common isotopes and ions.
• Construct dot diagrams, a shorthand notation for Bohr models, using the element symbol and dots to represent electrons in the
outermost energy level.
• Describe the charge, relative mass, and the location of protons, electrons, and neutrons within an atom.
• Calculate the number of protons, neutrons, electrons, and mass number in neutral atoms and ions.
• Explain how the different mass numbers of isotopes contributes to the average atomic mass for a given element (conceptual, no
calculations).
• Use isotopic notation to write symbols for various isotopes (ex. Carbon-12, C-12, 12C, etc.)
• Explain Bohr’s model of the atom.
• Draw Bohr models from hydrogen to argon including common isotopes and ions.
• Construct dot diagrams, a shorthand notation for Bohr models, using the element symbol and dots to represent electrons in the
outermost energy level.
Vocabulary
nucleus
Bohr model
proton
average atomic mass
electron
isotope
atomic number
energy levels
mass number
electron cloud
orbital
electron configuration
Bohr model
proton
average atomic mass
electron
isotope
atomic number
energy levels
mass number
electron cloud
orbital
electron configuration
Atomic Structure Jeopardy
https://jeopardylabs.com/play/atomic-structure156
jeopardylabs.com/play/atomic-structure156
jeopardylabs.com/play/atomic-structure156
Studying Atoms
Learning Targets
I can describe the historical development of atomic theory.
| studying_atoms.pptx | |
| File Size: | 1302 kb |
| File Type: | pptx |
History of the Modern Atomic Theory
Atomic Timeline
Atomic Models Jigsaw
| atomic_models_jigsaw_activity.pdf | |
| File Size: | 401 kb |
| File Type: | |
Structure of the Atom
Learning Targets
I can describe the subatomic particles in terms of their charge, relative mass, and location in an atom.
I can calculate the number of protons, neutrons, and electrons and the the mass number of an element or ion.
I can explain how isotopes are used to determine average atomic mass for an element.
I can calculate the number of protons, neutrons, and electrons and the the mass number of an element or ion.
I can explain how isotopes are used to determine average atomic mass for an element.
| structure_of_an_atom.pptx | |
| File Size: | 59 kb |
| File Type: | pptx |
| atomic_structure_notes.docx | |
| File Size: | 689 kb |
| File Type: | docx |
Isotopes and Average Atomic Mass
Learning Targets
I can differentiate between isotopes.
I can use isotopes to calculate the average atomic mass.
I can use isotopes to calculate the average atomic mass.
ChemTeam: Average Atomic Mass
Modern Atomic Theory
Learning Targets
I can explain and create a Bohr model diagram.
I can explain and create Bohr model diagrams for ions in elements.
I can explain and create Bohr model diagrams for ions in elements.
| modern_atomic_theory.pptx | |
| File Size: | 272 kb |
| File Type: | pptx |
The Bohr Model
PhET Simulation
https://phet.colorado.edu/en/simulation/hydrogen-atom
Firework Science
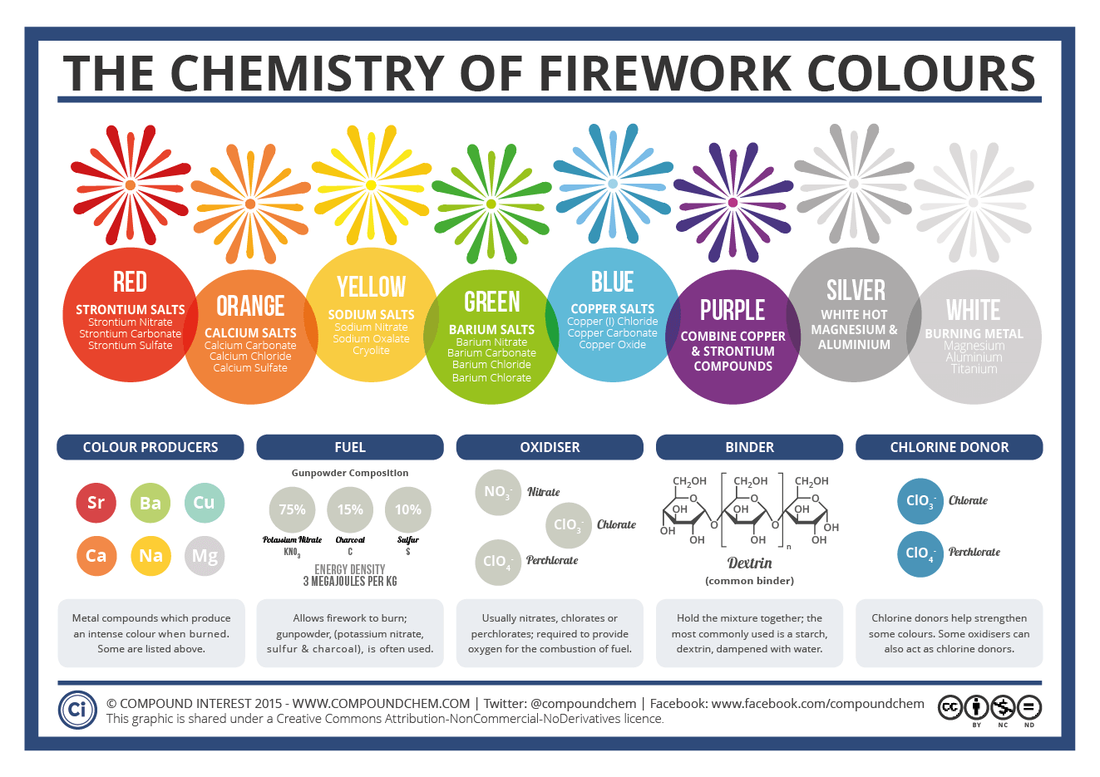
http://www.compoundchem.com/2013/12/30/the-chemistry-of-fireworks/